A Loss of Function,
Not a Gain of Toxicity
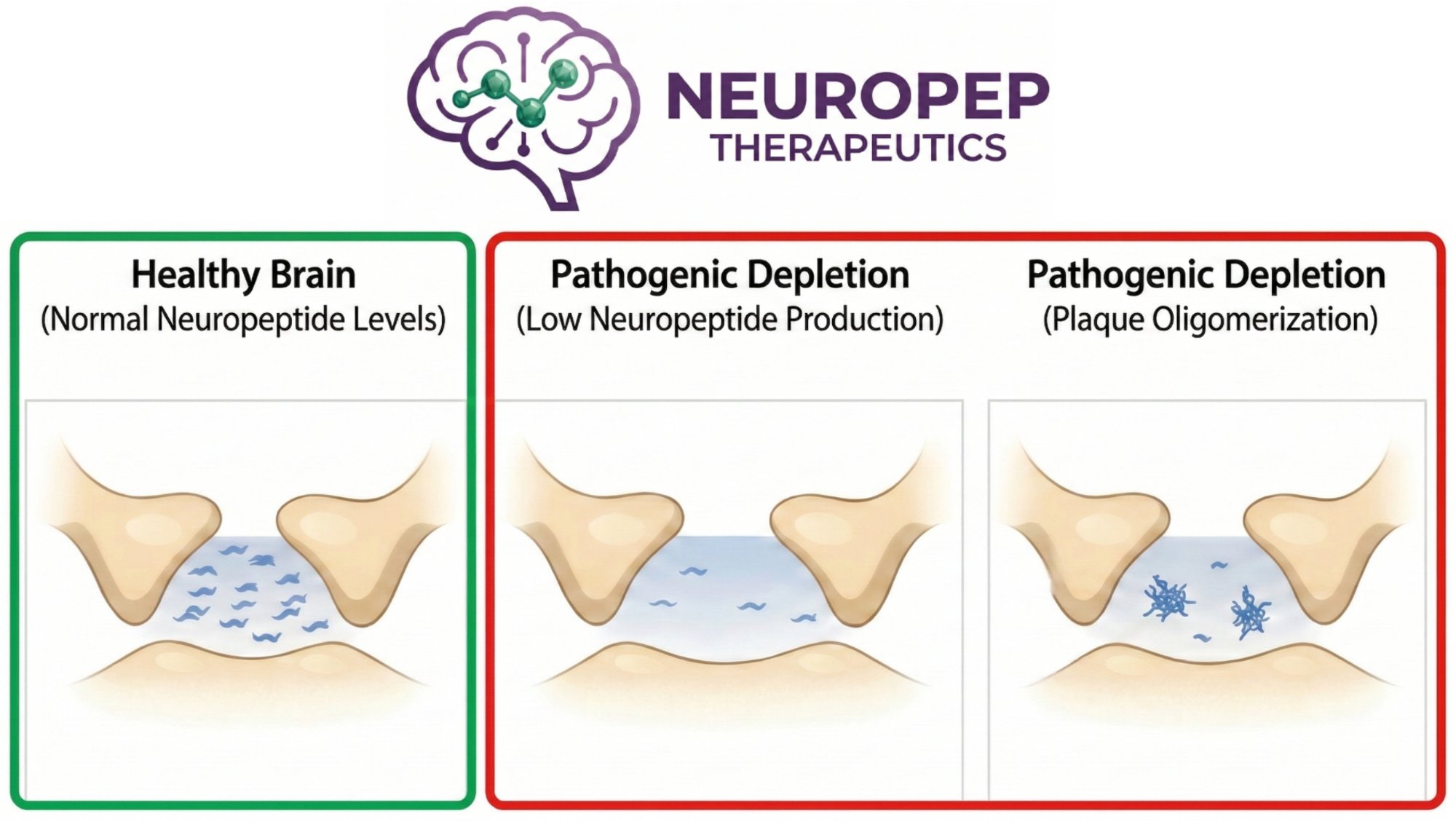
Even drugs that successfully reduced plaques by nearly 70% showed no cognitive improvement — and direct amyloid blocking drugs actually accelerated decline.
The breakthrough insight: Alzheimer's and Parkinson's are driven by neuropeptide depletion, not accumulation. When soluble neuropeptide levels fall below a critical threshold, synapses fail and cognition collapses.
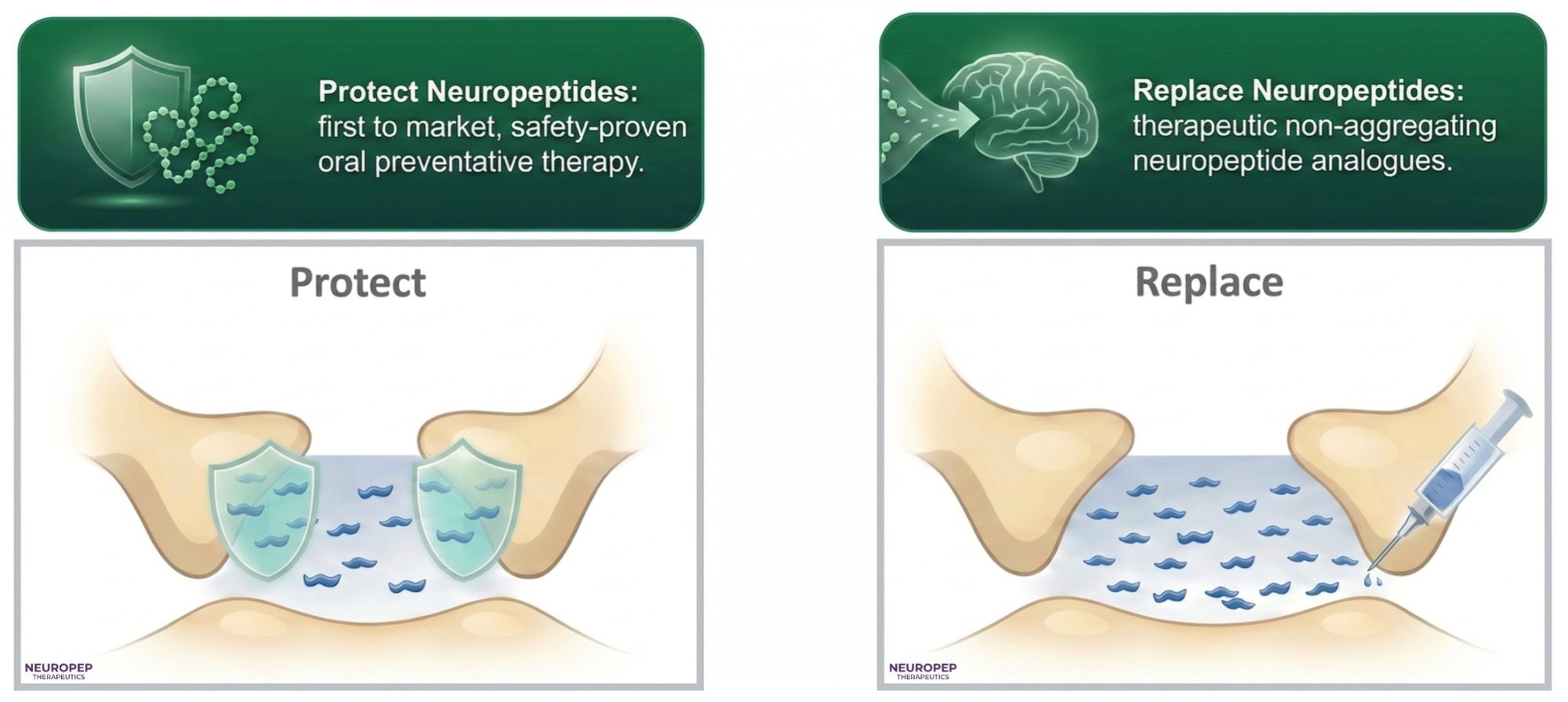
We restore what the brain needs.
Neuropep Therapeutics focuses on restoring synaptic function and recovering memory formation via two complementary approaches: protection of endogenous peptides in the CSF, and replacement via stable peptide analogues.